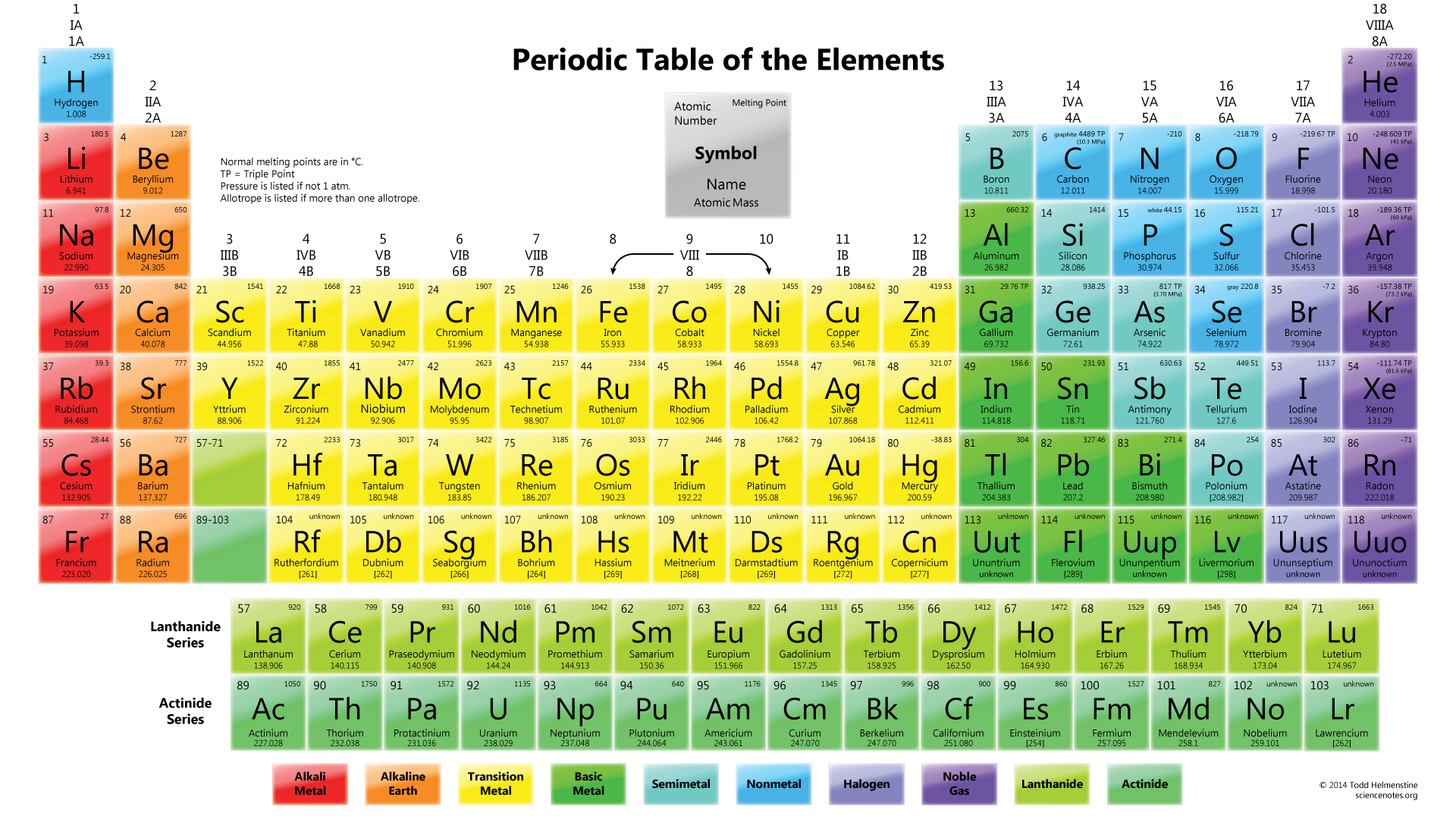
Thumbnail: Ionization energies superimposed on a periodic table. Chemistry Definition, History & Branches Related Study Materials. 2.S: Elements, Atoms, and the Periodic Table (Summary) To ensure that you understand the material in this chapter, you should review the meanings of the following bold terms and ask yourself how they relate to the topics in the chapter. Dmitri Mendeleev developed the first periodic table of elements with the 63 known elements identified at the time.2.E: Elements, Atoms, and the Periodic Table (Exercises) These are homework exercises to accompany Chapter 2 of the Ball et al.Some characteristics of the elements are related to their position on the periodic table. 2.7: The Periodic Table The chemical elements are arranged in a chart called the periodic table.2.6: Arrangements of Electrons Electrons are organized into shells and subshells about the nucleus of an atom.2.5: Atomic Masses Atoms have a mass that is based largely on the number of protons and neutrons in their nucleus. In chemistry, periodicity refers to repeating trends in element properties on the periodic table.Isotopes are atoms of the same element that have different masses. 2.4: Nuclei of Atoms Elements can be identified by their atomic number and mass number.Protons and neutrons are grouped together in the nucleus of an atom, while electrons orbit about the nucleus. 2.3: The Structure of Atoms Atoms are composed of three main subatomic particles: protons, neutrons, and electrons. 
The modern atomic theory establishes the concepts of atoms and how they compose matter. 2.2: Atomic Theory Atoms are the ultimate building blocks of all matter.The hydroxyl radical is very reactive, so it quickly reacts with other chemical species. The hydroxyl group is denoted by -OH in chemical structures and has a valence charge of -1. Chemical elements are represented by a one- or two-letter symbol. The hydroxyl group is a functional group consisting of a hydrogen atom covalently bonded to an oxygen atom. 2.1: The Elements All matter is composed of elements.Unprotected by enamel, a tooth will start to decay, thus developing cavities and other dental problems. Acids found in some foods or made by bacteria that feed on food residues on our teeth are capable of dissolving enamel. It has to be hard so that our teeth can serve us for a lifetime of biting and chewing however, tough as it is, tooth enamel is susceptible to chemical attack. 2.0: Prelude to Elements, Atoms, and the Periodic Table The hardest material in the human body is tooth enamel.Nonmetals are separated from metals by a line that cuts diagonally through the region of the periodic table containing elements with partially filled p orbitals.\) The nonmetals are located on the upper right side of the periodic table. Nonmetals include solids, liquids, and gases at room temperature and pressure.They tend not to be malleable or ductile, so they form brittle solids. Because the two words have defined different sets of elements over the years, the IUPAC. The term 'family' is synonymous with the term 'group'. Chemical families tend to be associated with the vertical columns on the periodic table. Nonmetals are typically poor conductors of heat and electricity, with low melting and boiling points. In chemistry, a family is a group of elements with similar chemical properties.For example, diamond and graphite are both pure carbon. Most nonmetals take many forms called allotropes.Nonmetals typically don't look metallic.The nonmetals include the group called the nonmetals, as well as the halogens and the noble gases.These elements are located on the righthand side of the table, with the exception of hydrogen.The nonmetals are a small group of elements on the periodic table.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
